CLINICAL REPORT
J. Prosthodont. 2026;35:235–242. wileyonlinelibrary.com/journal/jopr © 2025 by the American College of Prosthodontists.
Authors
Peter Rekawek DMD, MD1 Ariana Etessami DDS2 Frank Tuminelli DMD3 Lukasz Skomial DDS4 Gary Orentlicher DMD5
1Department of Dentistry, Division of Oral and Maxillofacial Surgery, New York University Langone Medical Center and Bellevue Hospital Center, New York, New York, USA
2Department of Dentistry, Division of Graduate Prosthodontics, New York Harbor Veterans Affairs Medical Center, New York, New York, USA
3Department of Dentistry, Division of Graduate Prosthodontics, New York Harbor Veterans Affairs Medical Center, New York, New York, USA
4Department of Dentistry, New York Harbor Veterans Affairs Medical Center, New York, New York, USA
5Department of Dentistry, Division of Oral and Maxillofacial Surgery, New York Harbor Veterans Affairs Medical Center, New York, New York, USA
Correspondence: Gary Orentlicher, DMD, Department of Dentistry, Department of Veterans Affairs, New York Harbor Medical Center, 423 East 23 Street, New York, NY 10010–5011, USA. Email: gary.orentlicher@va.gov; drgaryo@yahoo.com
Abstract
Alveolar bone morphology drives the edentulous patient’s rehabilitation. In edentulism, maxillary resorption is often in three vectors: superiorly, medially, and posteriorly, leading to a prosthetically unfavorable pseudo-class III ridge relationship. Longstanding edentulism, especially in the context of ill-fitting dentures, can result in severe maxillary atrophy. Rehabilitation of the severely atrophic maxilla remains a challenge, frequently requiring extensive bone grafting procedures in combination with endosseous dental implants. Graftless options, using longer endosseous and/or tilted dental implants placed into the nasal, pyriform rim, pterygoid process, and zygomatic bone, have been developed to solve this treatment dilemma. These options have specific patient anatomy limitations, can be technique sensitive, and can result in increased patient morbidity. Digitally planned, custom-milled, and rigidly fixated subperiosteal implants offer a recently introduced immediate fixed option when conventional solutions fail or are contraindicated. Cone-beam computed tomography (CBCT) and computer-aided design and computer-aided manufacturing (CAD-CAM) redefine a controversial 70-year-old concept, using prosthodontic reverse-engineering to offer stability, reduced morbidity, and improved patient satisfaction. Long-term follow-up for the use of this technique in fixed definitive restorations is not available at this time.
KEYWORDS: atrophic maxilla, computer-aided design, dental implant, full-arch reconstruction, subperiosteal implantation
Introduction
Alveolar bone volume, location, and morphology play a key role in the rehabilitation of the edentulous patient. In the presence of teeth or dental implants, occlusal force distribution, load, and muscle function help maintain alveolar bone volume.1 Edentulous maxillary resorption is in three vectors: superiorly, medially, and posteriorly. Many times, this leads to a prosthetically unfavorable pseudo-class III ridge relationship.2,3 When edentulism is experienced for extended time frames, coupled with poorly fitting dentures, the result is a severely atrophic maxilla.4–6 Rehabilitation of atrophic maxillae with dental implant therapy remains a challenge. Prior to dental implant placement, conventional treatment commonly requires staged procedures involving various bone grafting procedures, autogenous and/or allogeneic. Multiple procedures may be indicated, prolonging treatment time and patient morbidity. As a result, various “graftless” treatment protocols have been developed and introduced (i.e., All-on-Four Treatment Concept7 and Trans-nasal,8 Trans-sinus,9 Pterygoid,10,11 and Zygomatic implants).12,13 If a patient exhibits Cawood and Howell class V–VI bone atrophy, for whom conventional solutions have failed or are contraindicated, few options are available.14 Additionally, cone-beam computed tomography (CBCT) to examine the zygomatic bone can result in the discovery of inadequate bone thickness to predictably place a zygomatic implant. No surgical solution is without potential risks and complications. The above graftless treatment protocol’s potential complications are well documented.
This paper outlines the historical basis of subperiosteal implants, their development, and improvements made throughout the years. The contemporary reintroduction of the concept of this form of implant utilization, along with a suggested treatment planning and workflow, is discussed.
Each patient received a patient-specific, prosthetically driven, subperiosteal implant with an immediate fixed provisional (Individual Patient Solution, Preprosthetic [IPS], KLS Martin, Tuttlingen, Germany), as initially documented by Rahlf et al., Gellrich et al., and Korn et al.15–17 CBCT, three-dimensional (3D) virtual surgical planning (VSP), prosthodontic reverse-engineering, and computer-aided design and computer-aided manufacturing (CAD-CAM) were utilized to re-engineer a controversial 70-year-old concept. This resulted in the development of the IPS subperiosteal implant option.18–21
The IPS treatment was Food and Drug Administration (FDA) cleared in the USA in June 2022. The first USA patient case treatment plan was done in August 2023. The first treatment was planned in November 2023, and the surgery was performed in January 2024. To the date of this article’s submission, seven maxillary surgeries and one mandibular surgery have been performed, all treated with fixed provisional and planned definitive (final) restorations. Although only recently FDA cleared, this treatment has been successfully used in Europe for over 10 years with removable overdenture definitive prostheses.15–17,22–26 None of the subperiosteal implants with fixed provisional immediate loading have failed in this short follow-up period.
The first subperiosteal implant was placed in 1942 by Dahl, who pioneered the concept in Sweden.27 Goldberg and Gershkoff later introduced the concept to the United States in 1949.28 The initial attempts at subperiosteal implants were rudimentary and faced issues due to compromised technique and suboptimal materials. After its inception, efforts were made to refine implant parameters such as design and materials, all of which contributed to advancements in dental implantology. The original subperiosteal implants were placed in the mandible due to an abundance of basal bone.29,30 Since the maxillary alveolus consisted largely of cancellous bone, providing a poorer foundation for subperiosteal implant placement, maxillary subperiosteal implants were much less commonly placed.31
Different materials and surface treatments were attempted in the construction of the subperiosteal implant, with varying results reported in the literature. Vitallium, a cobalt-chromium alloy, was the first and best-known material used.32 In 1979, Leake et al. reported on carbon-coated implants with favorable results.33 In 1982, Hess et al. also attempted to coat the implants in carbon.34 They documented histological evidence of immune cell infiltration and immunological rejection of the carbon coating. Other researchers tested the efficacy of hydroxyapatite-coated subperiosteal implants with varying results.35
Although initially garnering interest, the original subperiosteal implant was fraught with challenges and poor clinical results. These were a result of invasive impression techniques, high incidence of infection, failure, and difficulty with implant positioning and stabilization.36 Most subperiosteal implants rested on the alveolar bone, under the tissue flaps, not secured to the underlying bone. As a result, the use of subperiosteal implants was progressively and largely abandoned. Simultaneously, the emergence of Branemark’s endosteal implants and the osseointegration concept shifted the treatment choice.37
The recent concept of subperiosteal implants has changed with the introduction of newer technologies and the use of titanium. The clinician now has use of precise CBCT/CT scan data, digital planning that is based on prosthesis design, CAD-CAM, strong thin biocompatible materials, precision milling of guides, stronger customized implants with intimate adaptation to the jawbone, and an ability to visualize the ideal areas for rigid fixation implant stabilization. The restoration that is fabricated is patient-specific, biocompatible, and rigidly stable, designed based on an ideal prosthodontic workflow. Immediate loading presents the clinician and patient with another option in a fixed dental restoration for edentulous areas with severe bone resorption.
VSP can be used to plan for a removable/overdenture definitive (final) restoration or for a fixed provisional/definitive restoration.38 The planning, design, and surgery will vary based on whether a removable or fixed definitive restoration is planned. Many subjects receiving this treatment have been treated for the reconstruction of discontinuity defects from benign and malignant tumor resections or congenital deformities, while the remainder have been performed for severely atrophic patients.15–17,22–26,39 The subperiosteal implant treatment presented in this report was restored with a fixed provisional restoration. The definitive restoration will be fixed as well.
The IPS subperiosteal implant and stabilization screws were made of Ti-6Al-4V titanium, like the material used to produce plates and screws for rigid fixation in orthognathic surgery, jaw fracture stabilization, and jaw resection surgeries. The implant does not follow the concepts and principles of osseointegration. The treatment follows the biomechanical principles of rigid fixation for the treatment of congenital or developmental dentofacial deformities, jaw fracture management, and benign and malignant tumor ablation surgery reconstruction.40–44 No human or animal histological evidence exists that shows osseointegration attachment between the implant and the underlying bone it rests on. Bone growth over and around the bone plates and support screws has been clinically and radiographically observed in the other applications noted above.45–47
Case Report
The prosthodontic and surgical evaluations and workups were similar for all patients. Medical histories were reviewed to rule out patients with contraindications to surgery and general anesthesia. Patients were evaluated clinically and radiographically to determine the severity of maxillary atrophy and the volume of their zygomatic bone. Clinical evaluations included assessment of their available inter-arch distance and restorative space, cervical-gingival smile line, residual ridge architecture, volume of attached gingiva, and identification of any tissue deformities or pathologies. Diagnostic CBCT scans were obtained to evaluate bone volume and further examine for pathology.
If deemed a good candidate, the patient must have well-functioning, well-adapted, and clinically acceptable complete dentures, either previously fabricated or newly fabricated for the planned treatment. Dentures must have exhibited patient-appropriate and patient-specific esthetics (tooth morphology, tooth setup, and lip support), proper phonetics (sibilant, fricative, and bilabial nasal sounds), and occlusion (vertical dimension and occlusal scheme). As per the dual-scan CBCT protocol, the denture was either duplicated in clear resin or directly used as a radiographic guide after fiducial markers were placed (i.e., Suremark [Suremark Inc., Mesa, AZ] or gutta percha). Seven to eight markers, in total, spread around the denture, were placed on the buccal and palatal/lingual surfaces of each prosthesis for the first CBCT scan. The patient was then scanned wearing these dentures, with an occlusal registration interposed between the arches for added position accuracy and stability. A second CBCT scan of the dentures alone was made, with the fiducial marker positions unchanged and no occlusal registration.
Digital Imaging and Communication in Medicine (DICOM) data sets were then created from the CBCT scans. The prosthesis was then digitized using an intraoral scanner (Trios 4, 3Shape, Copenhagen, Denmark). These scans were then converted into standard triangulation language (STL) format data sets. STL data sets are important to create ideal tooth morphology in the provisional restoration. The data sets were merged into a 3D rendering, which served as an underlying blueprint for the design of the patient-specific subperiosteal implant. A web-based VSP session served as the platform for multidisciplinary planning with the surgeon, prosthodontist, and the subperiosteal implant designer using Geomagic Freeform Plus software (OQTON Inc., Leuven, Belgium). Based on the imported planned provisional and definitive restoration position data, the team designed bone reduction cutting guides (if needed), the subperiosteal implant framework, including the number, heights, and locations of the pillars, and the numbers and locations of the stabilization screw holes. A screw-retained provisional restoration was fabricated for delivery at surgery. Key prosthodontic parameters steering the prosthesis design included proper anterior-posterior spread (A-P spread), prosthetically favorable pillar emergence, cervical-gingival smile-line, and minimizing the anterior and posterior cantilevers. Additional factors such as adequate bone reduction for restorative space, prosthesis strength, minimizing flanges, and maximizing oral hygiene access were also considered.
Surgical Evaluation
Partially edentulous patients were advised that all remaining teeth and implants were to be removed 6–8 weeks prior to surgery, allowing for complete soft tissue healing without openings in the crestal tissue.48 This also allowed for flexibility in incision design and favorable soft tissue closure over the subperiosteal implant and the pillars extending through the soft tissue into the mouth. Prior to surgery, written informed consent was obtained for both the surgical procedure and photographs.
Since dual-scan DICOM data sets were available, the data sets were imported into third-party proprietary guided surgery implant planning software (i.e., DTX [NobelBiocare, Kloten, Switzerland], Simplant [Dentsply Sirona, Charlotte, NC, USA], and coDiagnostiX [Straumann Group, Basel, Switzerland]) to further evaluate the patient case prior to the VSP treatment planning session.
When planning for a rehabilitation with a fixed prosthesis, using All-on-X surgical planning protocols is strongly recommended. Bone reduction concepts, anterior-posterior (A-P) spread, smile-line evaluation, and a minimum of 13–15 mm restorative space should be strongly considered.
VSP/Framework Design
Initially, the subperiosteal frameworks were designed to incorporate five pillars. As the process evolved, planning involved the use of four pillars to minimize soft tissue openings and reduce the likelihood of soft tissue dehiscence. It may be necessary to incorporate five pillars if the alveolar ridge is wide transversely or if a large anterior-posterior cantilever is present. Four pillars provide equal anterior-posterior spread and stability for a fixed prosthesis. Pillar positions are designed for emergence to be at the cingulum of the lateral incisor or canine sites and mid-occlusally at the second premolar or first molar sites. If using a fifth pillar, it is typically placed to emerge through the cingulum of one of the central incisors. The subperiosteal implant has integrated standard multiunit abutments (MUA) milled at the end of each pillar (i.e., NobelBiocare). This must be considered when determining restorative space and VSP requirements.
Some may find it uncomfortable in severe atrophy patient cases to reduce the alveolar bone height additionally. In many full-arch reconstruction patient cases, including digitally designed and fabricated fixed subperiosteal implants, bone reduction may be needed for esthetic and prosthodontic reconstruction considerations, and occasionally for surgical reasons. During VSP sessions, integrated bone reduction guides can be planned to ensure bone reduction accuracy.
The anatomy of the ridge should be considered as well. Tuberosity reductions are frequently necessary for additional vertical space to accommodate the posterior aspect of the prosthesis. Sinus exposure and possible Schneiderian membrane perforation may be inevitable during posterior bone reduction. Based on incision design, sinus perforations should heal without complication if adequate soft tissue availability and coverage are present in the dissected mucoperiosteal flaps.
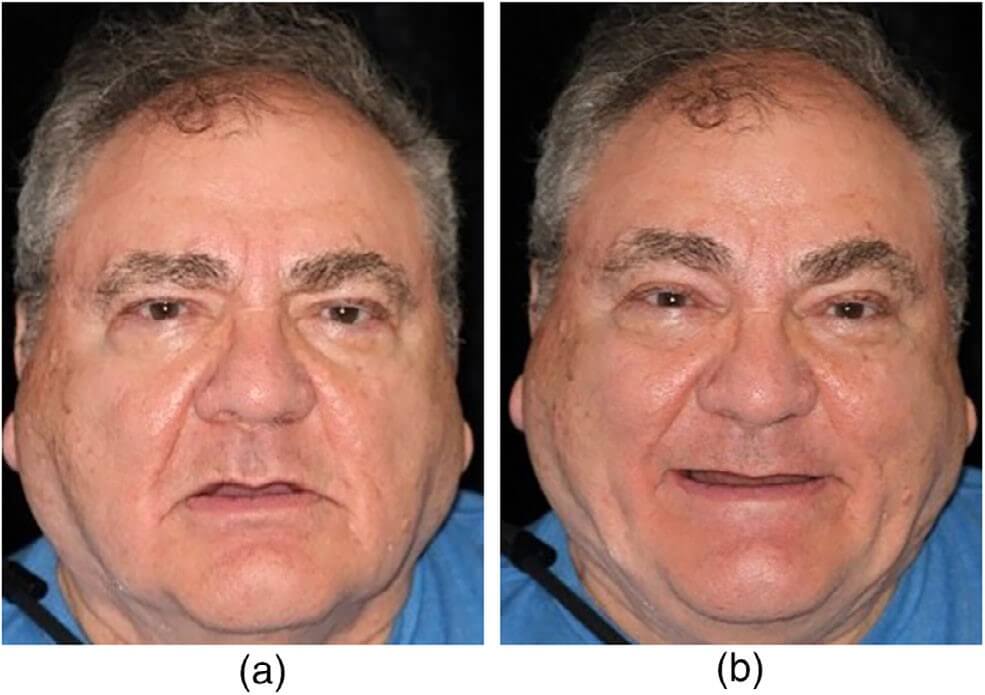
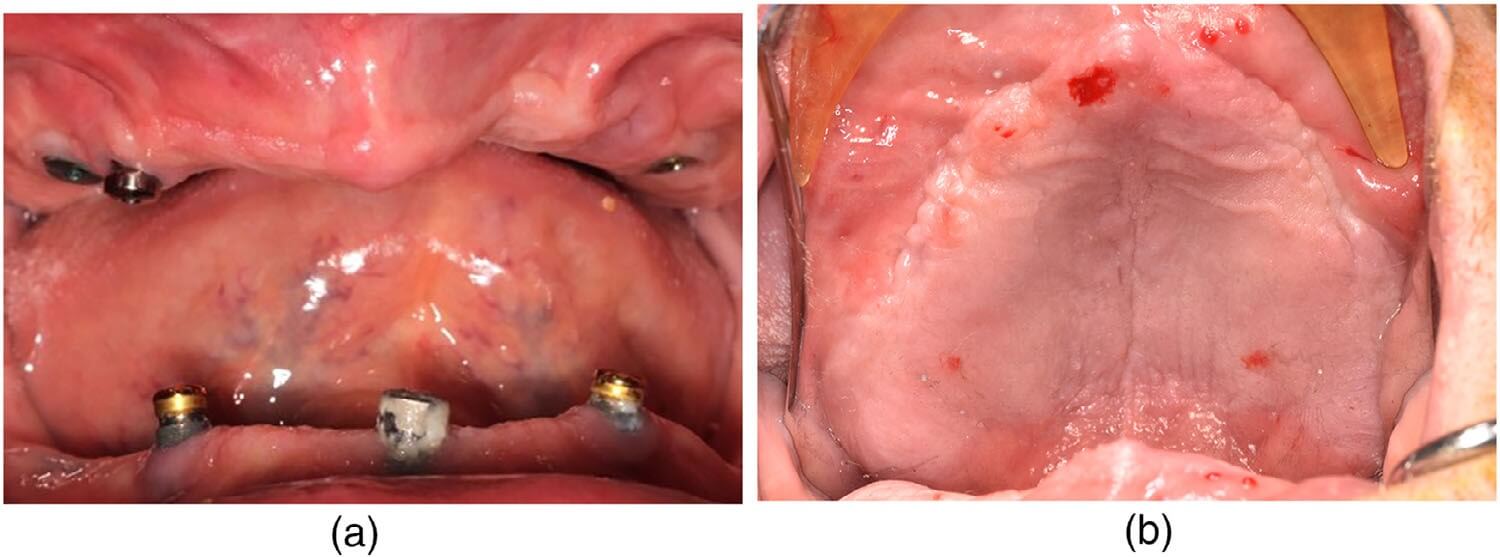
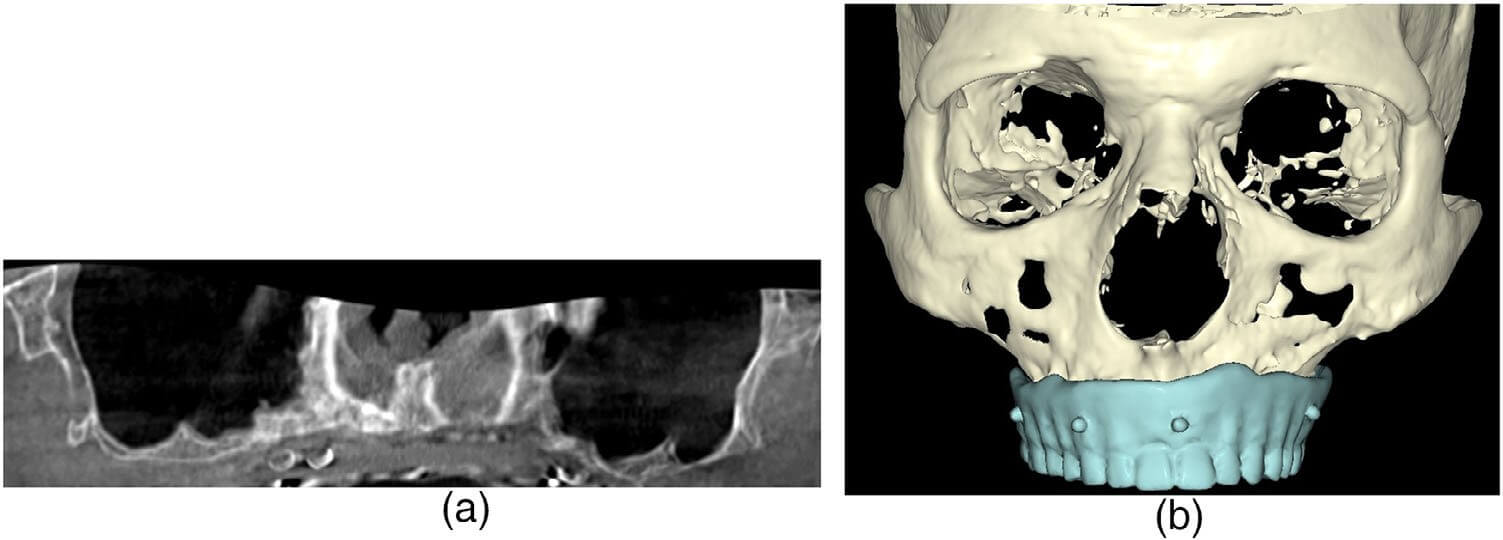
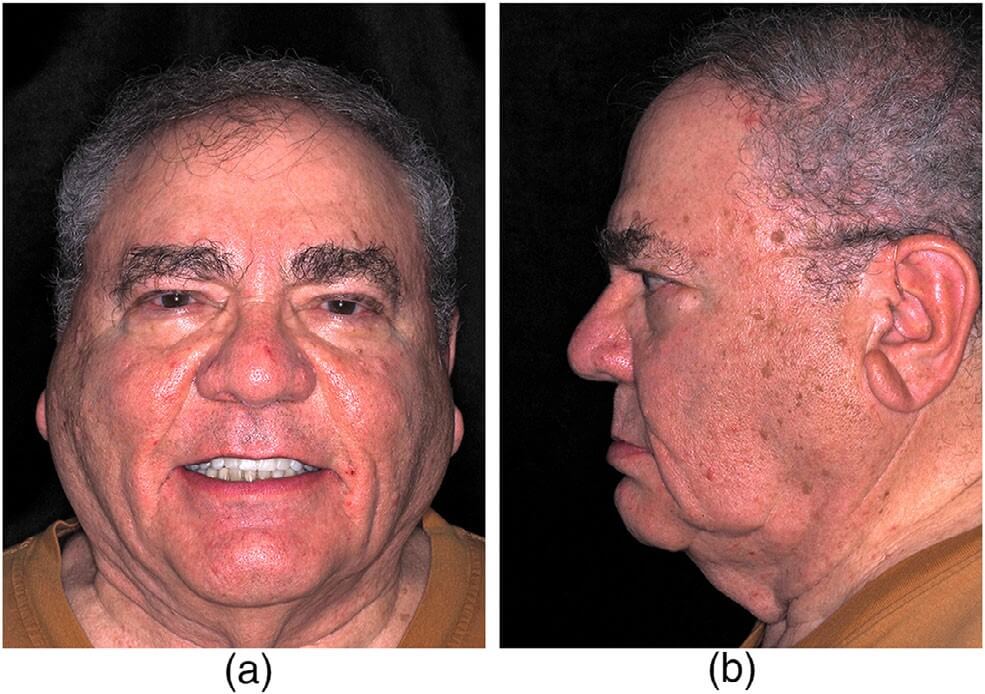
A 79-year-old male presented to the NY Harbor Veterans Affairs Medical Center Oral and Maxillofacial Surgery department as a referral from the Prosthodontic department. The patient had a past medical history significant for hypertension, peripheral vascular disease, and obstructive sleep apnea. The patient was edentulous, with 3 failing and fractured previously placed dental implants and severe atrophy of his maxilla (Figures 1a,b and 2a,b). He had difficulty functioning and tolerating a complete maxillary denture. A clinical and radiographic work-up with prosthodontic records and a full-head CBCT was performed as outlined previously (Figure 3a). The CBCT data was evaluated in the DTX software (NobelBiocare). This exam revealed severe vertical and horizontal atrophy of his maxillary alveolus with narrow, thin zygomatic bones (Figure 3b).
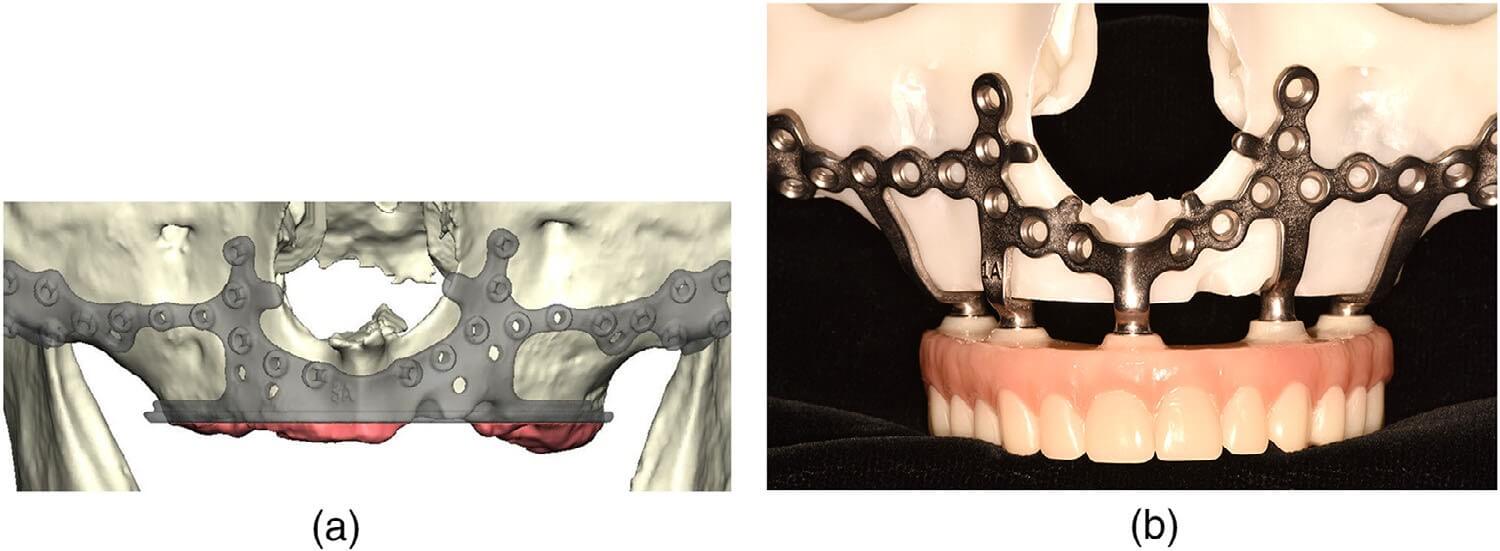
As per the IPS protocol, the appropriate DICOM and STL data sets were uploaded to the subperiosteal implant designers. A VSP was conducted with the designers and the prosthodontic and oral and maxillofacial surgery teams. The plan was approved by the teams, and the bone reduction guide, subperiosteal implant, and provisional restoration were fabricated and delivered prior to surgery (Figure 4a,b). Prior to surgery, titanium cylinders (NobelBiocare) were secured to the pillars of the subperiosteal implant and appropriately luted in place to the provisional restoration (Locator Chairside Attachment Processing Material, Zest Dental, Carlsbad, CA, USA). The intaglio surface of the provisional restoration was then trimmed and polished for insertion immediately after surgery.
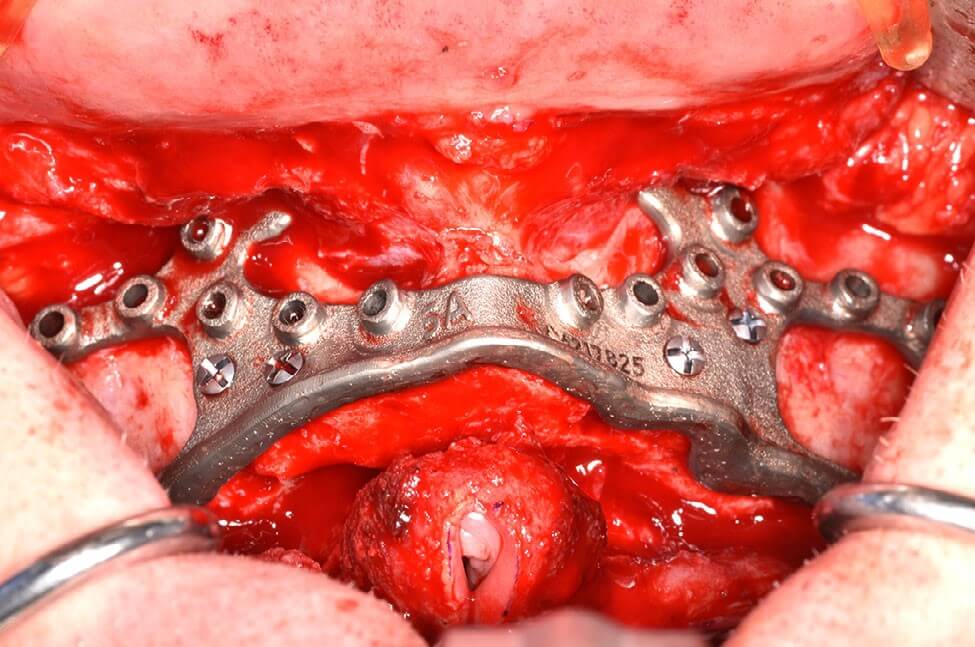
The surgery was performed under general anesthesia with a nasoendotracheal intubation in the main operating room. In standard oral and maxillofacial surgical fashion, a high vestibular incision was made using a thin, pointy electrocautery tip, avoiding the infraorbital nerves. Standard surgical exposure of the maxilla was performed from malar buttress to malar buttress, including the bilateral nasal pyriform rims and floor, bilateral infraorbital nerves, and palatal alveolus. Care was taken to remove all soft tissue fragments attached to the maxillary bone in the areas that the subperiosteal implant would rest on. A titanium bone reduction guide was placed appropriately using four integrated pyriform rim tabs and secured to the maxilla with eight 2.0 titanium screws. Bone reduction was performed with an 8 mm oval egg-shaped bur (Stryker Corporation, Kalamazoo, MI, USA) under copious normal saline irrigation (Figure 5).
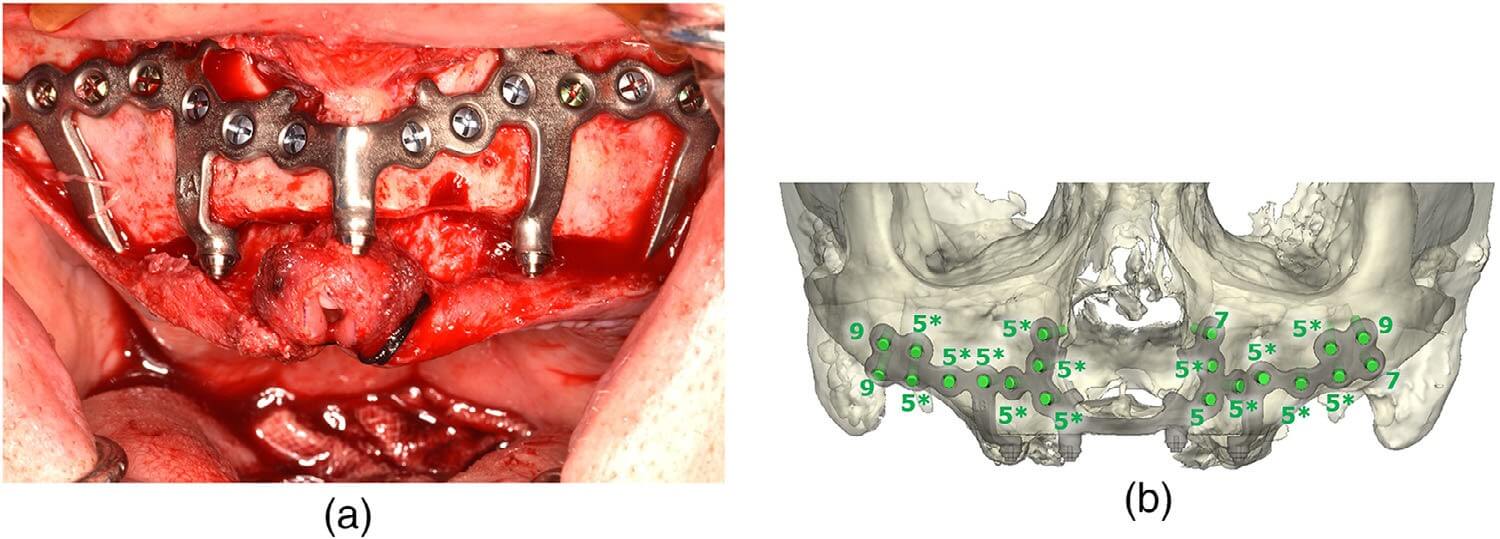
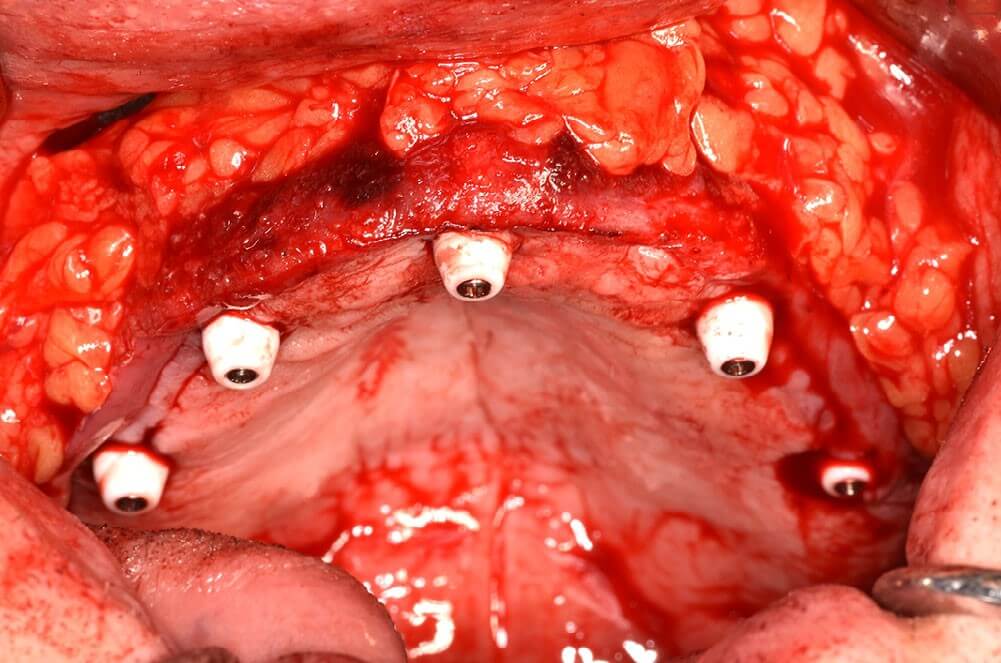
The bone reduction guide was removed, and the subperiosteal implant was then placed using the same four integrated pyriform rim tabs and secured passively to the maxilla using twenty 2.0 mm locking titanium screws of 5–9 mm premeasured lengths, as per the surgical plan. The longer screws placed in the bilateral malar buttress and pyriform regions were bicortical. The remaining screws were unicortical in the lateral maxilla (Figures 6a,b). Bilaterally, using blunt surgical dissection, the buccal fat pads were exposed and advanced over the secured implant plate and sutured in place anteriorly with Vicryl sutures. Tissue punches were used to create openings for perforation of the pillars through the tissue flaps (Figure 7). The wounds were then closed with resorbable chromic gut sutures in a running fashion. The provisional restoration was then screwed into the subperiosteal implant pillars with standard M1.4 MU abutment prosthodontic screws (NobelBiocare) for immediate loading (Figure 8a,b).
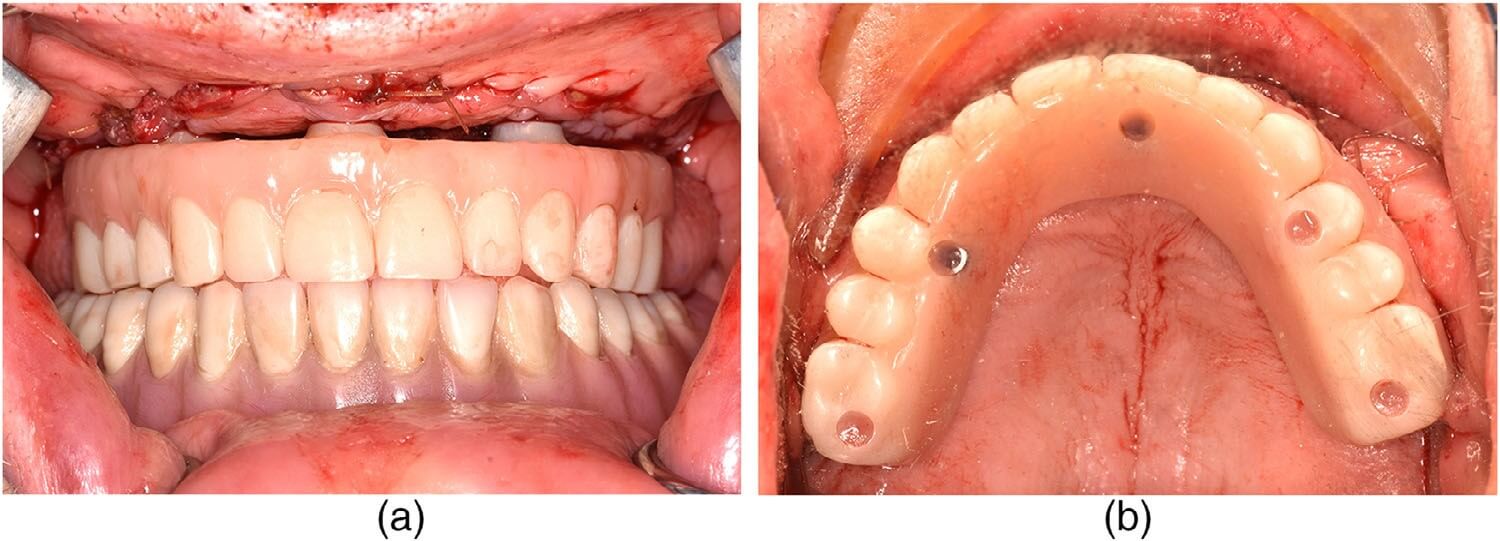
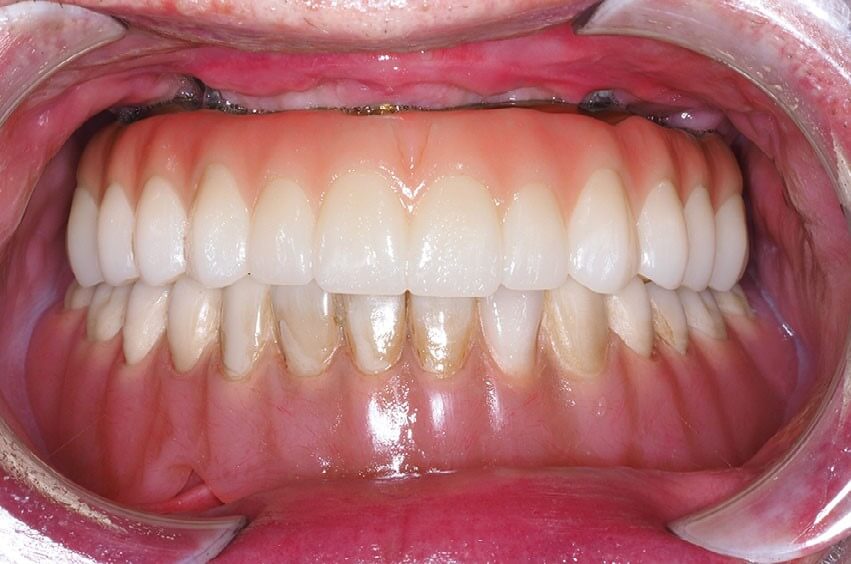
The patient was prescribed amoxicillin, ibuprofen, acetaminophen, oxycodone, and chlorhexidine at discharge on the same day as the surgery. Three days after surgery, the patient was seen for imaging and follow-up. The healing was appropriate, with postoperative edema and pain consistent with the procedure. All incisions were intact with no signs of infection or wound dehiscence. The patient was seen weekly for the first 4 weeks, then monthly until 3 months postoperatively. At the 1-, 2-, and 3-month follow-up visits, all soft tissue was healed appropriately with no dehiscence, and edema and bruising had completely subsided. He was maintaining a soft diet, and the provisional restoration was intact and functioning well. There was no evidence of infection. At the 3-month postoperative visit, standard prosthodontic records were made for fabrication of the definitive restoration. A monolithic zirconia full arch implant-supported prosthesis on a titanium bar substructure was inserted at 4.5 months postoperatively (Figures 9 and 10a,b).
Summary
It is important to note that this technology is not without its own prosthodontic and surgical nuances. Careful planning, high-quality records, and an integrated prosthodontic/surgical team approach for decision-making during the VSP planning session are required. Intraoperative surgical design choices, from a prosthodontic and surgical perspective, ultimately dictate the success of the implant and prosthesis. This reduces patient morbidities and complications.
Only short-term follow-up statements can be made at this time. There have not been any postoperative infections in these patient cases. Potential postoperative complications include infection, mucositis, soft-tissue breakdown and dehiscence around the pillars perforating the mucosa, and wound breakdown resulting in plate exposure, as well as prosthodontic complications inherent in full-arch fixed hybrid reconstructions. Close continued postoperative follow-up is necessary to observe for these problems. Patients should be encouraged to maintain good oral hygiene and prosthodontic follow-up.
With existing digital technologies, CAD-CAM, biocompatible materials, and well-established prosthodontic and surgical principles, the Individual Patient Solution, Preprosthetic (IPS), is a new adaptation of a well-known old technology. It may provide the prosthodontic and surgical team with a new solution to successfully treat a difficult subgroup of severely atrophic patients with a low level of potential morbidity and complications. IPS is potentially a promising treatment option, and early results of this technology are encouraging. However, the lack of long-term follow-up with this procedure in severely atrophic patients desiring a fixed provisional and definitive restoration requires further investigation.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflicts of interest.
Disclosure Statement
This material is the result of work supported with resources and the use of facilities at the New York Harbor Veterans Affairs Medical Center, New York, NY, USA. Its contents do not represent the views of VA or the United States Government.
ORCID
Frank Tuminelli DMD https://orcid.org/0000-0003-1292-7017
Gary Orentlicher DMD https://orcid.org/0000-0002-3461-1753
References
- Khalifa AK, Wada M, Ikebe K, Maeda Y. To what extent can residual alveolar ridge be preserved by implants? A systematic review. Int J Implant Dent. 2016;23(2):22.
- Reyes A, Serret L, Peguero M, Tanaka O. Diagnosis and treatment of pseudo-class III malocclusion. Case Rep Dent. 2014;2014:652936.
- Ngan P, Moon W. Evolution of class III treatment in orthodontics. Amer J Orthod Dentofac Orthoped. 2015;148:22–26.
- Usui M, Onizuka S, Sato T, Kokabu S, Ariyoshi W, Nakashima K. Mechanism of alveolar bone destruction in periodontitis: Periodontal bacteria and inflammation. Jpn Dent Sci Rev. 2021;13:201–8.
- Alsaggaf A, Fenlon MR. A case control study to investigate the effects of denture wear on residual alveolar ridge resorption in edentulous patients. J Dent. 2020;98:103373.
- Kovacic I, Celebic A, Zlataric DK. Decreasing residual height in complete denture wearers. A five year follow up study. Coll Antropol. 2010;34:1051–56.
- Soto-Penaloza D, Zaragozi-Alonso R, Penarrocha-Diago M, Penarrocha-Diago M. The All-on-four treatment concept: Systematic review. J Clin Exp Dent. 2017;9:e474–88.
- Almeida P, Cacciacane SH, Ayrton AJ. Extra-long transnasal implants as alternative for Quad Zygoma. Ann Med Surg (Lond). 2021;27:102635.
- Aalam AA, Krivitsky-Aalam A, Zelig D, Oh S, Holtzclaw D, Kurtzman GM. Trans-sinus dental implants, for immediate placement when insufficient alveolar height is present: An alternative to zygomatic implants—surgical case series. Ann Med Surg (Lond). 2023;85:51–56.
- Balaji VR, Lambodharan R, Manikandan D, Deenadayalan S. Pterygoid implant for atrophic posterior maxilla. J Pharm Bioallied Sci. 2017;9:S261–63.
- Tulasne JF. Implant treatment of missing posterior dentition. In: Albrektsson T, Zarb GA, editors. The Branemark osseointegrated implant. Chicago: Quintessence; 1989. p. 103.
- Maló P, Araujo NM, Lopes I. A new approach to rehabilitate the severely atrophic maxilla using extramaxillary anchored implants in immediate function: A pilot study. J Prosth Dent. 2008;100:354–56.
- Aparicio CE. Zygomatic implants: the anatomy-guided approach (illustrated ed.). Chicago: Quintessence; 2012.
- Cawood JI, Howell RA. A classification of edentulous jaws. Int J Oral Maxillofacial Surg. 1988;17:232–36.
- Rahlf B, Korn P, Zeller A-N, et al. Novel approach for treating challenging implant-borne maxillary dental rehabilitation cases of cleft lip and palate: A retrospective study. Int J Implant Dent. 2002;8(1):6.
- Gellrich N-C, Korn P, Jehn P, Neuhaus M, Lentge F, Rahlf B. Exceptional cases demand exceptional personalized solutions: the next level in dental rehabilitation. J Pers Med. 2024;14:294.
- Korn P, Gellrich N-C, Jehn P, Spalthoff S, Rahlf B. A new strategy for patient-specific implant-borne dental rehabilitation in patients with extended maxillary defects. Front Oncol. 2021;11:718872.
- Efanov JI, Roy AA, Huang KN, Borsuk DE. Virtual surgical planning: the pearls and pitfalls. Plast Reconstr Surg Glob Open. 2018;6(1):e1443.
- Portnoy Y, Koren J, Khoury A, et al. Three-dimensional technologies in presurgical planning of bone surgeries: current evidence and future perspectives. Int J Surg. 2023;109(1):3–10.
- Velarde K, Cafino R, Isla A, et al. Virtual surgical planning in craniomaxillofacial surgery: A structured review. Comput Assist Surg. 2023;28(1):2271160.
- Singh GD, Singh M. Virtual surgical planning: modeling from the present to the future. J Clin Med. 2021;10(23):5655.
- Korn P, Gellrich N-C, Spalthoff S, et al. Managing the severely atrophic maxilla: Farewell to zygomatic implants and extensive augmentations? J Stomatol Oral Maxillofac Surg. 2022;123:562–65.
- Gellrich N-C, Zimmerer RM, Spalthoff S, et al. A customized digitally engineered solution for fixed dental rehabilitation in severe bone deficiency: A new innovative line extension in implant dentistry. J Cranio Max Fac Surg. 2017;45:1632–38.
- Gellrich N-C, Rahlf B, Zimmerer R, Pott P-C, Rana M. A new concept for implant-borne dental rehabilitation; how to overcome the biological weak-spot of conventional dental implants? Head Face Med. 2017;13(1):17.
- Jehn P, Spalthoff S, Korn P, et al. Oral health-related quality of life in tumour patients treated with patient-specific dental implants. Int J Oral Maxillofac Surg. 2020;49(8):1067–72.
- Spalthoff S, Borrmann M, Jehn P, Rahlf B, Gellrich N-C, Korn P. Comparison of conventional and digital workflow for dental rehabilitation with a novel patient-specific framework implant system. Int J Implant Dent. 2022;8(1):4.
- Dahl GSA. Om mojligheten for implantation i de kaken an metallskelett som bas eller retention for fosta eller aotaglosa protester. Odontologisk Tidskrift. 1943;51:440–49.
- Goldberg N, Gershkoff A. The implant lower denture. Dent Dig. 1949;55:490–94.
- Dantas T, Vaz P, Samuel F. Subperiosteal dental implants: past or future? A critical review on clinical trials/case reports and future directions. J Dent Implant. 2023;13:35.
- Markiewicz MR, Nishiyama K, Yago K, et al. Draining orocutaneous fistula associated with a failing subperiosteal implant: report of a case. J Oral Implantol. 2007;33:347–52.
- Demirdjan E. The complete maxillary subperiosteal implant: An overview of its evolution. J Oral Implantol. 1998;24:196–97.
- Weinberg BD. Subperiosteal implantation of a vitallium (cobalt-chromium alloy) artificial abutment. J Am Dent Assoc. 1950;40:549–62.
- Leake D, Reed OK, Armitage J, Bokros J, Haubold A. Carbon-coated subperiosteal dental implants for fixed and movable prostheses. J Prosth Dent. 1979;42:327–34.
- Hess JA, Molinari JA, Mentag PJ. Two cases of incompatibility to carbon-coated subperiosteal implants. Oral Surg Oral Med Oral Pathol. 1982;54:499–505.
- Golec TS. The use of hydroxylapatite to coat subperiosteal implants. J Oral Implantol. 1985;12:21–39.
- Strappa EM, Memè L, Cerea M, Roy M, Bambini F. Custom-made additively manufactured subperiosteal implant. Minerva Dent Oral Sci. 2022;71:353–60.
- Buser D, Sennerby L, De Bruyn H. Modern implant dentistry based on osseointegration: 50 years of progress, current trends and open questions. Periodontol 2000. 2017;73:7–21.
- Probst FA, Liokatis P, Mast G, Ehrenfeld M. Virtual planning for mandible resection and reconstruction. Innov Surg Sci. 2023;8:137–48.
- Vatteroni E, Covani U, Menchini Fabris GB. The new generation of subperiosteal implants for patient-specific treatment of atrophic dental arches: literature review and two case reports. Int J Periodontics Restor Dent. 2023;43:735–41.
- Bottlang M, Schemitsch CE, Nauth A, et al. Biomechanical concepts for fracture fixation. J Orthop Trauma. 2015;12(0 12):S28–S33.
- Yadav A. Principles of internal fixation in maxillofacial surgery. In: Bonanthaya K, et al., editors. Oral and maxillofacial surgery for the clinician. Singapore: Springer; 2021.
- Lovald ST, Wagner JD, Baack B. Biomechanical optimization of bone plates used in rigid fixation of mandibular fractures. J Oral Maxillofac Surg. 2009;67(5):973–85.
- Savoldelli C, Ehrmann E, Tillier Y. Biomechanical assessment of different fixation methods in mandibular high sagittal oblique osteotomy using a three-dimensional finite element analysis model. Sci Rep. 2021;11(1):8755.
- Kummer FJ. Craniomaxillofacial bone healing, biomechanics, and rigid internal fixation. In: Greenberg AM, Prein J, editors. Craniomaxillofacial reconstructive and corrective bone surgery. New York: Springer; 2002.
- Pacifici L, DE Angelis F, Orefici A, Cielo A. Metals used in maxillofacial surgery. Oral Implantol (Rome). 2017;9(1–4):107–11.
- Payne KFB, Balasundaram I, Deb S, Di Silvio L, Fan KFM. Tissue engineering technology and its possible applications in oral and maxillofacial surgery. Br J Oral Maxillofac Surg. 2014;52(1):7–15.
- Sahoo NK, Anand SC, Bhardwaj JR, Sachdeva VP, Sapru BL. Bone response to stainless steel and titanium bone plates: an experimental study on animals. Med J Armed Forces India. 2017;50(1):10–14.
- Hämmerle CH, Giannobile WV. Working Group 1 of the European Workshop on Periodontology. Biology of soft tissue wound healing and regeneration—consensus report of group 1 of the 10th European Workshop on Periodontology. J Clin Periodontol. 2014;41(Suppl 15):S1–S5.
How to cite this article: Rekawek P, Etessami A, Tuminelli F, Skomial L, Orentlicher G. A digitally designed fixed subperiosteal implant solution for the treatment of the severely atrophic full-arch with an immediate-load protocol. J Prosthodont. 2026;35:235–42. https://doi.org/10.1111/jopr.70071
